- Fact Sheet: Pre-Exposure Prophylaxis for HIV Prevention
- Fact Sheet – PrEP During Conception, Pregnancy, and Breastfeeding
Reports
- Estimated Percentages and Numbers of Adults with Indications for Preexposure Prophylaxis to Prevent HIV Acquisition — United States, 2015, Morbidity and Mortality Weekly Report (MMWR)
- Increased Medicaid Prescriptions for Preexposure Prophylaxis Against HIV infection — New York, 2012–2015, Morbidity and Mortality Weekly Report (MMWR)
En Español
Videos
PrEP Consultation Service for Clinicians
1-855 HIV-PREP (1-855-448-7737)
11 a.m. – 8 p.m. ET, Monday-Friday
For more information on the services offered through the PrEPline, visit the National Clinicians Consultation Center.
Free Continuing Education for Healthcare Providers
Advancing PrEP in Practice: Practical Strategies for Everyday Challenges
This activity is intended for primary care physicians, ID/HIV specialists, and public health and preventive medicine specialists. The goal of this activity is to improve recognition among primary care providers of patients who could benefit from human immunodeficiency virus (HIV) pre-exposure prophylaxis (PrEP) medications.
Faculty: Colleen F. Kelley, MD, MPH
Preventing HIV Infection in the Primary Care Setting: The Role of Pre-Exposure Prophylaxis (PrEP)
This CME activity can be completed through Medscape, and is intended for primary care physicians, ID/HIV specialists, nurses, and public health & preventive medicine specialists. The goal of this activity is to discuss the use of PrEP in the primary care setting, including patient assessment and management, CDC guidelines, and tips for incorporating this into practice. Requires free Medscape log in.
Moderator: Michael S. Saag, MD
Faculty: Demetre C. Daskalakis, MD, MPH; Christian B. Ramers, MD, MPH, AAHIVS; Dawn K. Smith, MD, MS, MPH
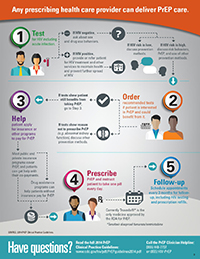
PrEP Guidelines
CDC has released a 2017 update to the clinical practice guidelines for PrEP. The new guidelines
- Provide clear criteria for determining a person’s HIV risk and indications for PrEP use.
- Require that patients receive HIV testing to confirm negative status before starting PrEP.
- Recommend regular monitoring of HIV infection status, side effects, adherence, and sexual or injection risk behaviors.
- Underscore importance of counseling about adherence and HIV risk reduction, including encouraging condom use for additional protection.
- The update to the providers’ supplement offers clinicians additional materials and tools for use when prescribing PrEP.
The guidelines recommend that PrEP be offered to people who are HIV-negative and at substantial risk for HIV.
For sexual transmission, this includes anyone who 1) is not in a mutually monogamous* relationship with a partner who recently tested HIV-negative, and 2) is a
- gay or bisexual man who has had anal sex without a condom or been diagnosed with an STD in the past 6 months; or
- heterosexual man or woman who does not regularly use condoms during sex with partners of unknown HIV status who are at substantial risk of HIV infection (e.g., people who inject drugs or have bisexual male partners).
For people in HIV-discordant couples where one sexual partner is HIV-positive and the other is HIV-negative. PrEP can be especially useful when the HIV-positive partner is not taking antiretroviral medications or does not have an undetectable viral load. Health care providers should also discuss the use of PrEP with HIV discordant heterosexual couples during conception and pregnancy as one of several options to protect the partner who is HIV-negative.
For people who inject drugs, this includes those who have injected illicit drugs in the past 6 months and who have shared injection equipment or been in drug treatment for injection drug use in the past 6 months.
Patients on PrEP should return to their health care provider every 3 months for a repeat HIV test and other follow-up. At this time, the provider can write a prescription refill, offer to counsel about medication adherence and risk reduction, test for STDs if necessary, and assess side effects.
PrEP is only for people who are at ongoing substantial risk of HIV infection. For people who need to prevent HIV after a single high-risk event of potential HIV exposure—such as unprotected sex, needle-sharing injection drug use, or sexual assault—there is another option called postexposure prophylaxis or PEP. PEP must begin within 72 hours of exposure. See our PEP guidelines at https://www.cdc.gov/hiv/pdf/programresources/cdc-hiv-npep-guidelines.pdf.
* Mutually monogamous means that you and your partner only have sex with each other and do not have sex outside the relationship.
Additional PrEP Resources
PrEP Clinical Trials
The guidelines are based on strong evidence from clinical trials of PrEP use in high-risk populations. All participants in these trials received pills containing either PrEP or placebo (a pill without any medicine in it), along with intensive counseling on safe-sex behavior, regular testing for STDs, and a regular supply of condoms.
In all of these studies, HIV transmission risk was lowest for participants who took the pill consistently. Specifically:
- Among gay and bisexual men, those who were given PrEP were 44% less likely overall to get HIV than those who were given a placebo. Among the men with detectable levels of medicine in their blood (meaning they had taken the pill consistently), PrEP reduced the risk of infection by as much as 92%. (iPrEx Study)
- Among heterosexually active men and women, PrEP reduced the risk of getting HIV by 62%. Participants who became infected had far less drug in their blood, compared with matched participants who remained uninfected. (TDF2 Study)
- Among men and women in HIV discordant couples, those who received PrEP were 75% less likely to become infected than those on placebo. Among those with detectable levels of medicine in their blood, PrEP reduced the risk of HIV infection by up to 90%. (Partners PrEP Study)
- Among injection drug users, a once-daily tablet containing tenofovir (one of the two drugs prescribed) reduced the risk of getting HIV by 49%. For participants who had detectable tenofovir in their blood, PrEP reduced the risk of infection by 74%. (Bangkok Tenofovir Study)
- None of the studies found any significant safety concerns with use of daily oral PrEP. Some trial participants reported side effects such as an upset stomach or loss of appetite but these were mild and usually resolved in the first month.